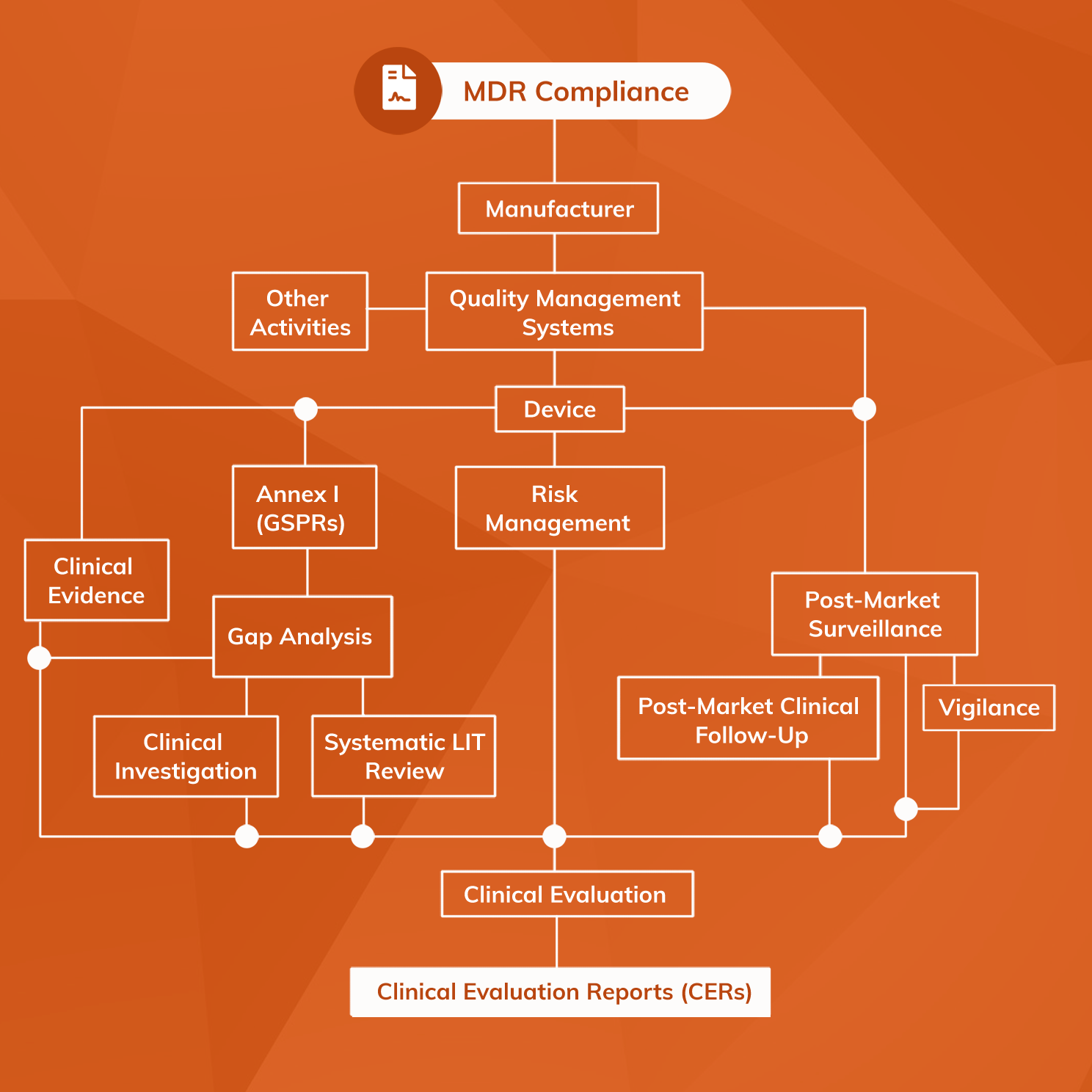
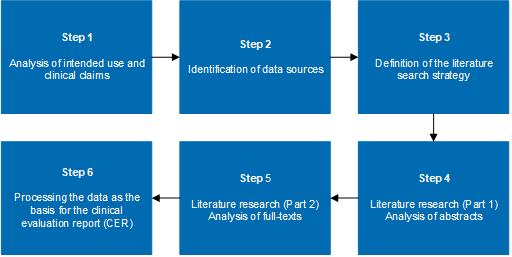
Clinical Evaluation Report for Medical Devices-Role of Medical Writer – Turacoz Healthcare Solutions

Clinical Evaluation Report for Europe and MEDDEV Expectations: Impact with New Medical Device Regulations (MDR) | Medical Events

Using a Clinical Evaluation Report template to write EU MDR-compliant CERs for your medical devices - YouTube