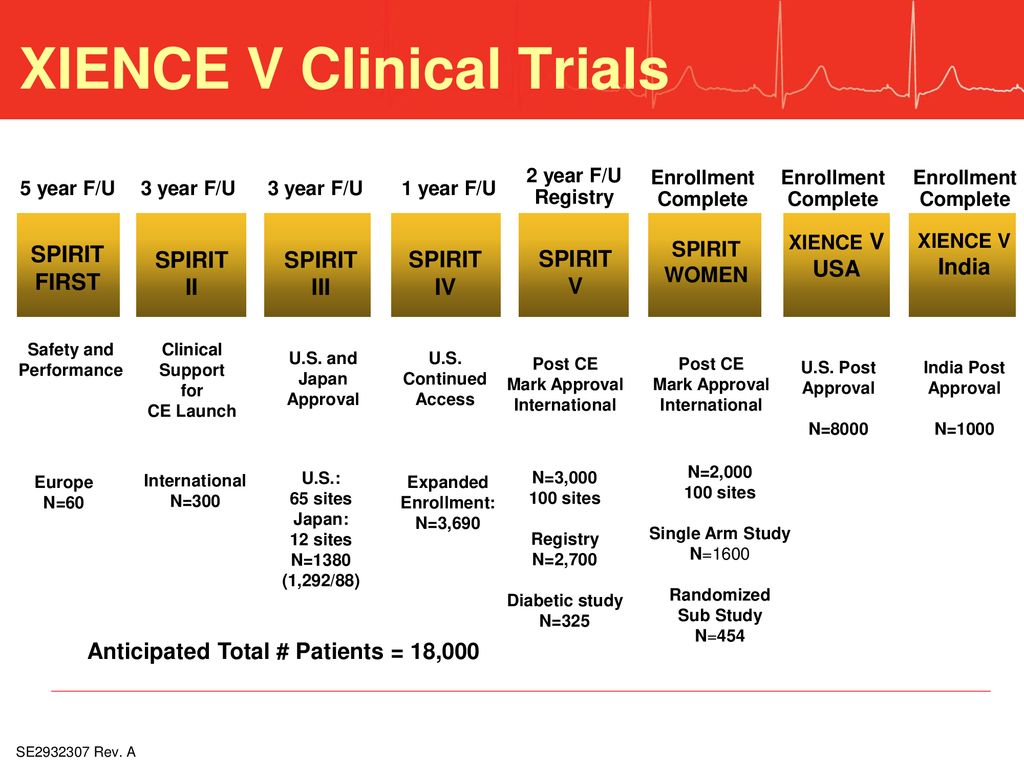
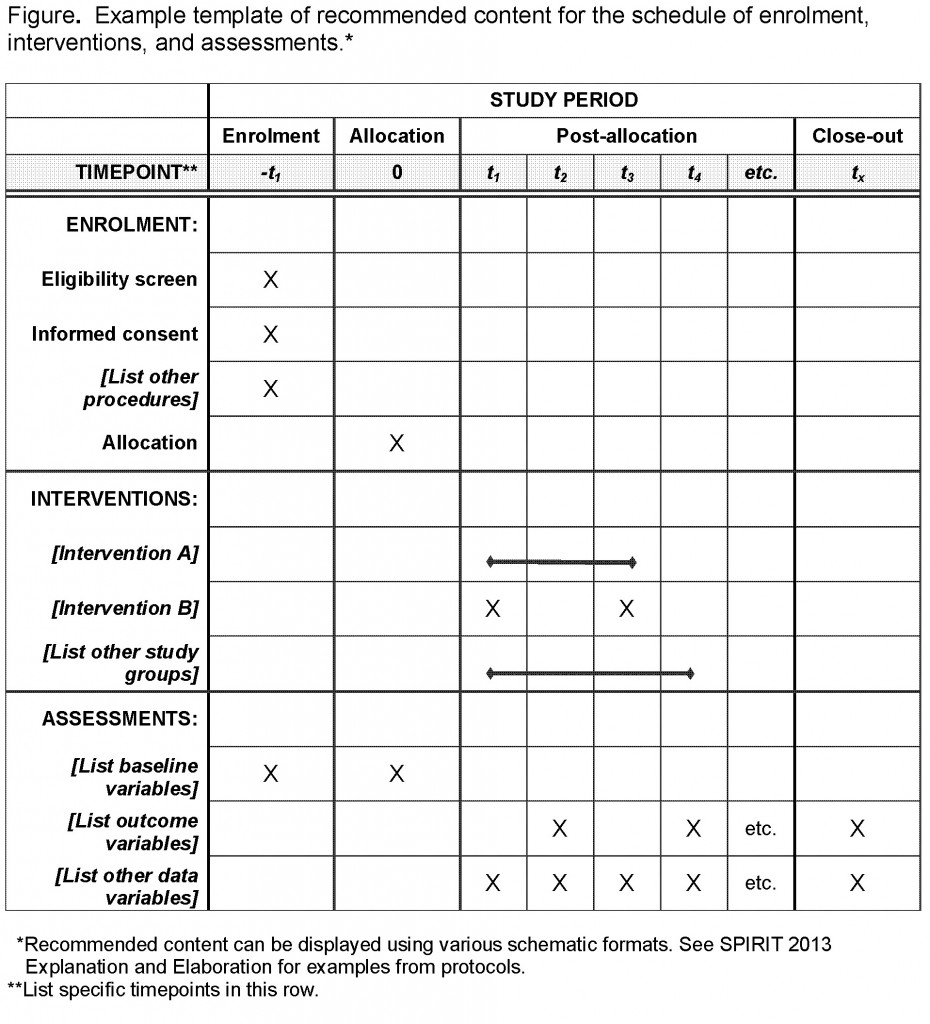
SPIRIT diagram. The figure details the timing of enrollment activities,... | Download Scientific Diagram

Table 2 from SPIRIT 2013 statement: defining standard protocol items for clinical trials. | Semantic Scholar
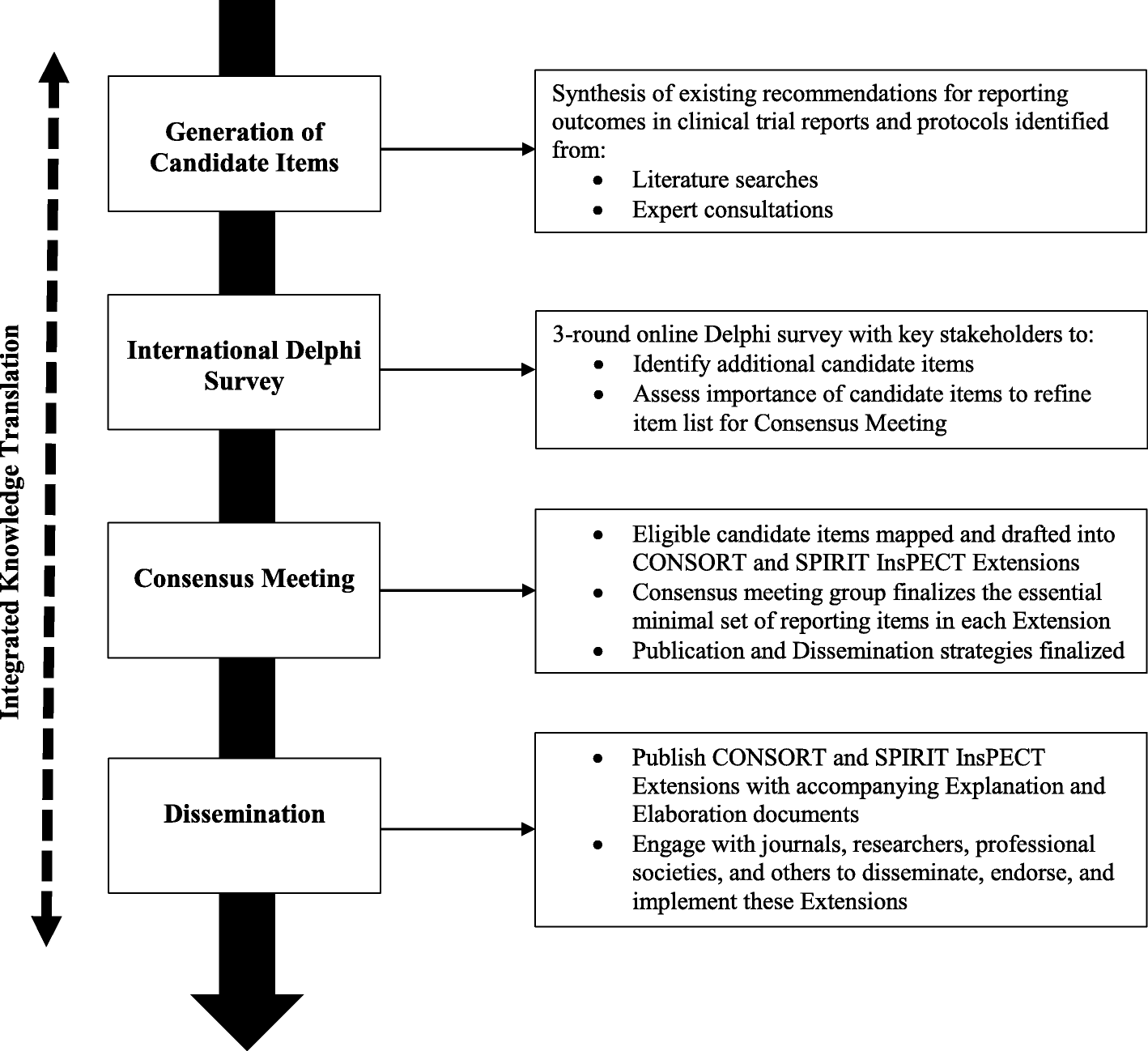
Improving outcome reporting in clinical trial reports and protocols: study protocol for the Instrument for reporting Planned Endpoints in Clinical Trials (InsPECT) | Trials | Full Text
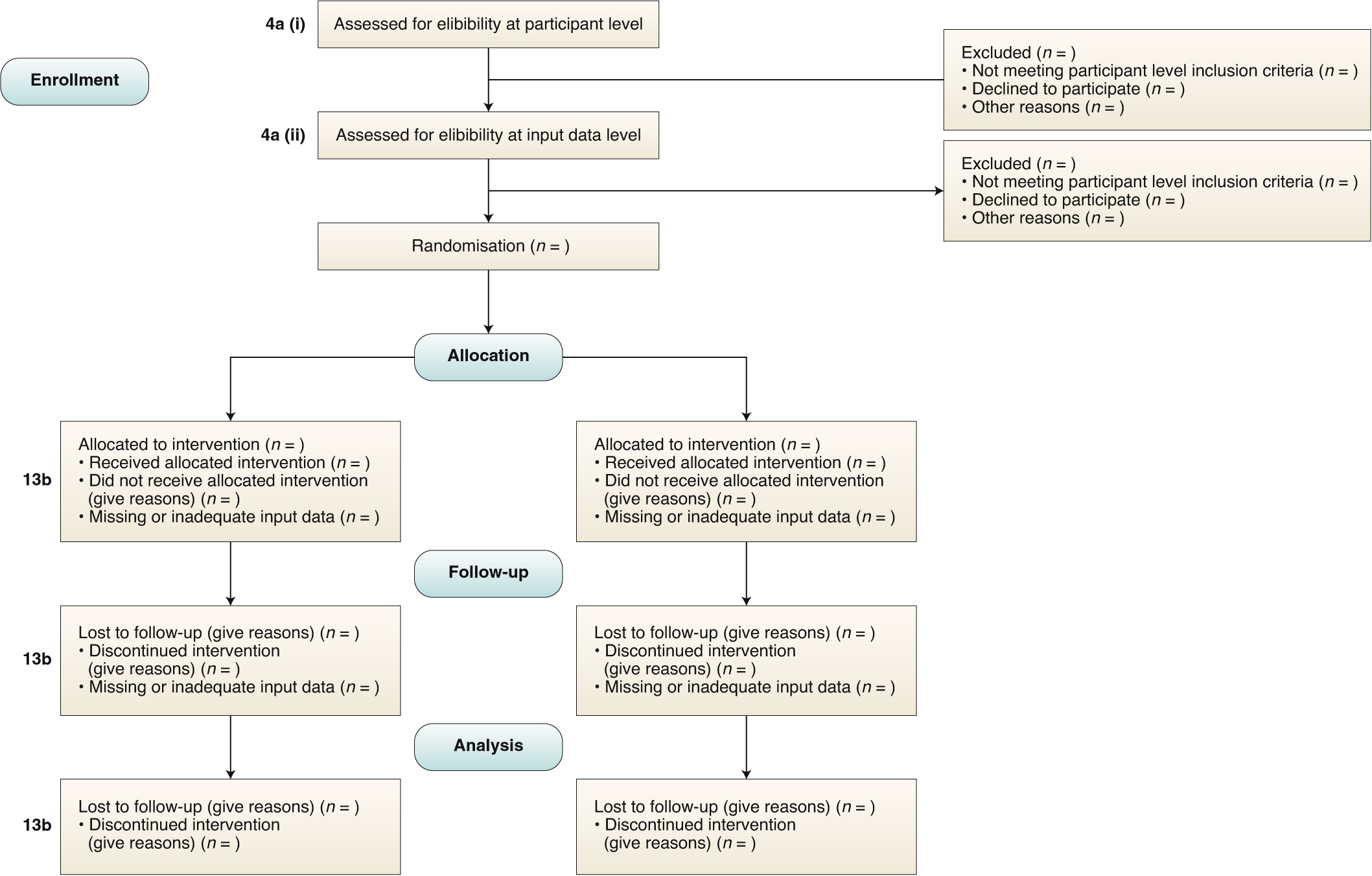
Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension | Nature Medicine
Guidelines for Inclusion of Patient-Reported Outcomes in Clinical Trial ProtocolsThe SPIRIT-PRO Extension

Guidelines for cellular and molecular pathology content in clinical trial protocols: the SPIRIT-Path extension - The Lancet Oncology

Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI Extension | The BMJ

PDF) SPIRIT 2013 Statement: Defining Standard Protocol Items for Clinical Trials | Trish Groves - Academia.edu

Guidelines for clinical trial protocols for interventions involving artificial intelligence: the SPIRIT-AI extension - The Lancet Digital Health

Reporting guidelines for clinical trial reports for interventions involving artificial intelligence: the CONSORT-AI extension - ScienceDirect