Phase I/II Trial of SPK-8011: Stable and Durable FVIII Expression for > 2 Years With Significant ABR Improvements in Initial Dose Cohorts Following AAV-Mediated FVIII Gene Transfer for Hemophilia A

PDF) A Phase 1/2 Trial of Investigational Spk-8011 in Hemophilia a Demonstrates Durable Expression and Prevention of Bleeds
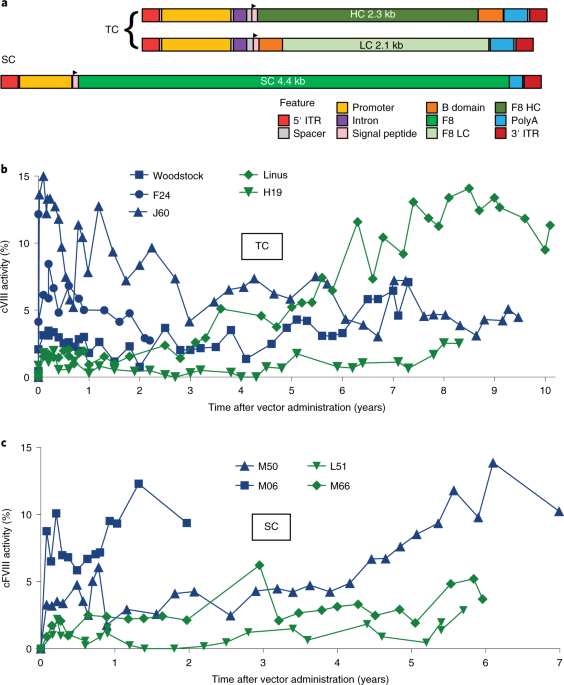
A long-term study of AAV gene therapy in dogs with hemophilia A identifies clonal expansions of transduced liver cells | Nature Biotechnology

Phase I/II Trial of SPK-8011: Stable and Durable FVIII Expression for > 2 Years With Significant ABR Improvements in Initial Dose Cohorts Following AAV-Mediated FVIII Gene Transfer for Hemophilia A

Preclinical assessment of an optimized AAV-FVIII vector in mice and non-human primates for the treatment of hemophilia A: Molecular Therapy - Methods & Clinical Development

The intersection of vector biology, gene therapy, and hemophilia - Lisowski - 2021 - Research and Practice in Thrombosis and Haemostasis - Wiley Online Library

From Clinical Trials to Clinical Practice: Practical Considerations for Gene Replacement Therapy in SMA Type 1 - Pediatric Neurology

Phase I/II Trial of SPK-8011: Stable and Durable FVIII Expression for >2 Years with Significant ABR Improvements in Initial Dose Cohorts Following AAV-Mediated FVIII Gene Transfer for Hemophilia A - ISTH Congress
Hemophilia A Clinical Gene Therapy Trials (September 2020). Listed are... | Download Scientific Diagram